While tissue engineering strategies for most organs stem primarily from a limited number of organ donors and the negative side effects of immunosuppressive treatments, the demand for a transplantable artificial ovary (TAO) emerges as a procedure for fertility restoration in cancer survivors, that cannot benefit from available fertility restoration solutions. Indeed, chemo- and radiotherapy can potentially be toxic for the ovaries, ultimately leading to sterility. In this context, the gynecology research team from the Catholic University of Louvain recently conducted the first attempt of “reverse engineering” the human ovary.

Image above: Emna Ouni, one of the researchers that carried out the study.
In biology, reverse engineering refers to the concept of taking apart a process or mechanism in order to understand it, re-engineer it (perhaps in a new way) and apply it to the biological world. In this project, the microenvironment of the human ovarian cell (extracellular matrix or ECM) was deconstructed to better understand its proteomic, architectural and mechanical cues and its role in ovarian activity from prepuberty until the menopause. The data uncovered will enable the TAO to be a future life-changer for many female patients, making motherhood a possibility after cancer survival.
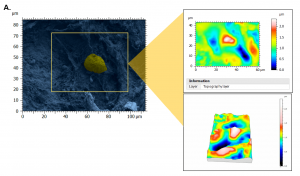
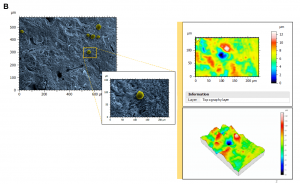
Figure 1. Topography of perifollicular ECM before and after menopause. SEM images were colorized with SMILE VIEW ™ Map software, based on MountainsSEM®, to highlight the presence of follicles (yellow) and distinguish them from the ECM (blue). Topography rendering shows a remodeling and difference in perifollicular ECM between reproductive age (A) and menopausal (B) tissues.
“If the cell is the basis of life, the extracellular matrix is the basis of multicellular life”. Based on this statement, Emna Ouni, PhD student on the UCL research team, has been an advocate of the role ECM plays in fertility for the last 5 years. In this study, she and her colleagues shed light on one of the ECM’s overlooked features: topography. The rationale for exploring the link between topography and fertility is that native ECM displays physical topography at the nanoscale and thus features of similar size on engineered substrates may be a better mimic. Furthermore, emerging in vitro studies demonstrate that ECM surface features play a role in a plethora of biological processes: cell differentiation, cell shape and morphology, cell adhesion, mechanotransduction, tumor propagation, dormancy and even ECM composition.
To provide the first description of these structural features, ovarian tissues from prepubertal, reproductive-age and menopausal patients were analyzed by scanning electron microscopy (SEM) at different magnifications to capture: fibril (nanoscale), fiber (microscale) and fiber-bundle (mesoscale) organization and diameters, as well as pore number and area, as a reflection of ECM network tightness. Based on a stereoscopic reconstruction of SEM captures created by tilting the sample by + 5° along the X-axis and generating 3D renderings using SMILE VIEW™ Map software based on MountainsSEM®, the team was able to extract surface topography and measure microroughness on the same tissue regions used to characterize ECM architecture.
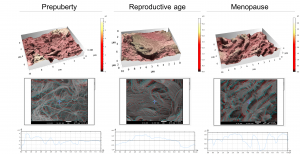
Figure 2. Architecture, topography and biomechanics synergy. 3D rendering of surface topography from stereoscopic reconstruction of two SEM images. Color scales indicate the height range on each surface. Scale on SEM micrographs corresponds to 1µm.
In the course of these analyses, the mechanical features of ovarian tissue recorded by atomic force microscopy (AFM) was linked to its nanotopography, ultrastructure and organization. The team discovered that soft ovarian tissue from reproductive-age patients was characterized by a significantly smoother tissue surface (99 nm ± 33) compared to the rougher prepubertal (228 nm ± 87) and menopausal (238 nm ± 28) tissues, which are more rigid. This points to a possible synergy between tissue rigidity and surface roughness.
Overall, in the human ovary, the team found that topography, in completion to biomolecular cues, synergizes with the ECM elasticity to influence ovarian tissue remodeling from prepuberty until menopause. Considering age as the most prominent fertility biomarker to date, the unique topological features that Ouni and her team succeeded in recording and reporting in their blueprint of the human ovarian ECM can be linked to female fertility, namely follicle activation and dormancy.
Above: Video animation of the 3D topography of ovarian tissue on reproductive age.
Moreover, the analyses demonstrated that ovarian ECM at reproductive age represents an ideal microenvironment for ovarian cell- and follicle- function and development due to its unique topological and mechanical features. Thus, the study provided the ECM cues necessary to replicate ovarian tissue properties in a functional engineered ovary and represents a reliable reference to compare physiological and pathological ovarian conditions based on ECM status.
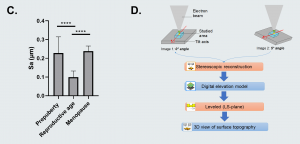
Figure C. Surface roughness measured by calculating the arithmetic average of 3D roughness (Sa) according to calculation conditions defined in ISO 25178. Figure D. 3D stereoscopic reconstruction workflow using SMILE VIEW™ Map software, based on MountainsSEM®. For full statistical details check the source article.
Instruments & software used
JEOL JSM-7500F field emission scanning electron microscope + SMILE VIEW™ Map software, based on MountainsSEM®.
Read more
Read the full article in Nature: https://rdcu.be/czVYd
About the author
Emna Ouni is a PhD bioengineer at the Gustave Roussy Institute.